Ideal Products for Clinical Trials and Commercialization
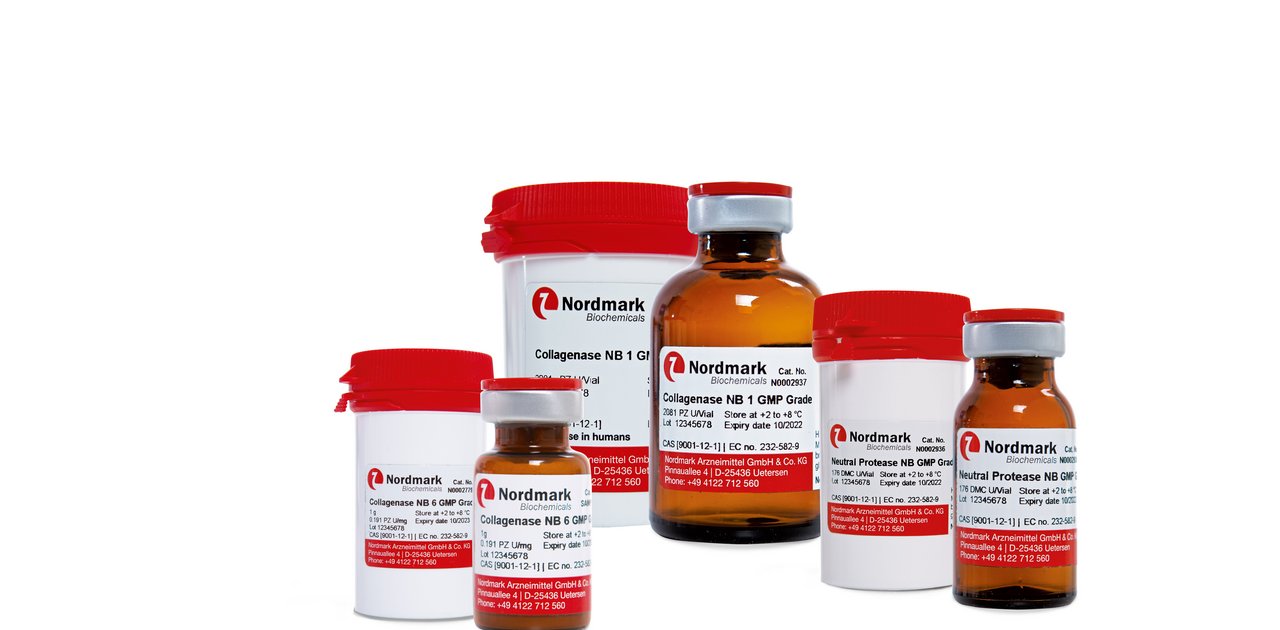
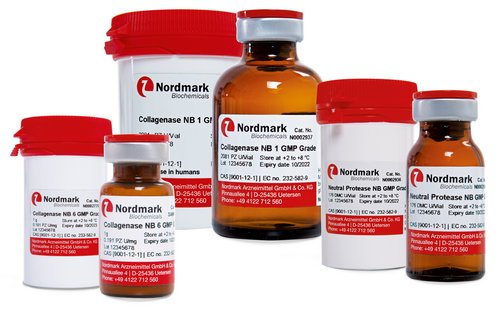
GMP Collagenase NB
Enzymes for tissue dissociation are critical raw materials for tissue engineering and transplantation into humans. Thus, Nordmark Biochemicals offers superior GMP grade Collagenase NB and Neutral Protease NB products that are manufactured in compliance with the EU-Guide to Good Manufacturing Practice (GMP). TSE safety of the products is certified and virus validation studies have been performed.
Superb quality and performance of our highest standard GMP Grade enzymes are assured by:
- stringent quality control
- reliable lot-to-lot consistency
- accurate analysis of proteolytic enzyme activities
- in-process controls
- manufacture according to GMP guidelines
Animal-Free GMP Collagenase
Animal-Free (AF) products are our superior enzymes designed to provide the highest possible safety for clinical applications. A plant-based production process and the use of carefully selected materials ensure that there is no commingling with components of animal origin. Thus, any risk of introducing potential animal-derived pathogens is avoided in the first place. Furthermore, the sophisticated production process ensures that there is no cross-contamination with any animal-derived materials. The animal-free enzymes are produced in compliance with GMP guidelines that guarantees the highest quality and a reliable lot-to-lot consistency. Therefore, these enzymes provide the highest level of safety and reduced regulatory hurdles.
Collagenase AF-1 GMP Grade and Neutral Protease AF GMP Grade represent the first choice for clinical isolation of especially sensitive cells like pancreatic islets, hepatocytes and dental pulp.
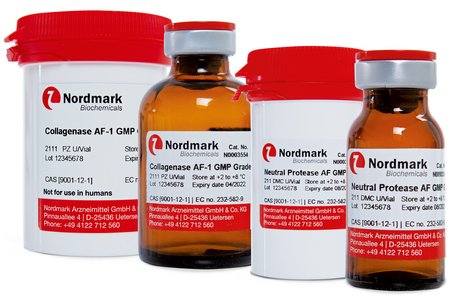
| Animal-free GMP Collagenase |
|---|
| Collagenase AF-1 GMP Grade |
| Neutral Protease AF GMP Grade |